GW Pharmaceuticals (NASDAQ: GWPH) has been on a tear over the past month after exceeding expectations with its Q4 results, which were released at the end of February.
GW By the Numbers
Revenue increased 68% year on year to $6.8 million, comfortably ahead of Wall Street’s anticipated $5.4 million. Net loss of $0.20 per share was in line with its performance the previous year, and again it beat the consensus estimate.
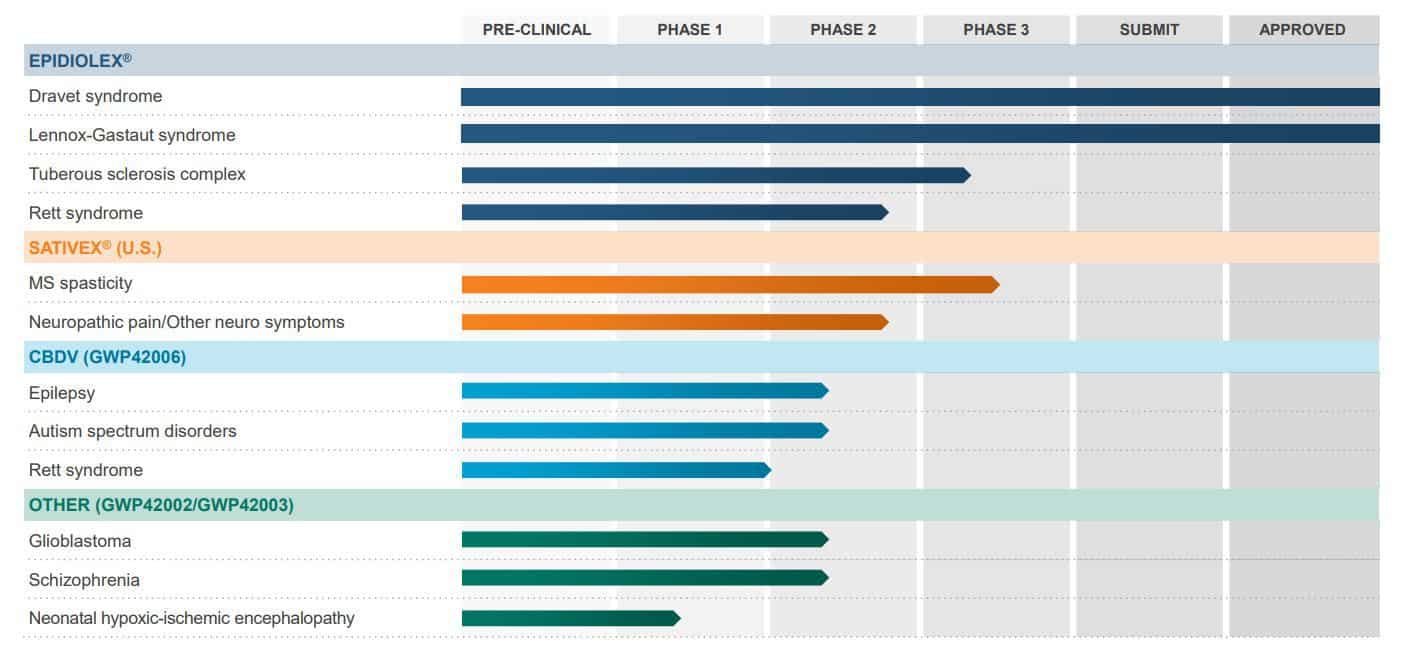
The operational highlight in Q4 was a strong launch of its flagship medicinal cannabis product, Epidiolex, in the US. It is the first CBD drug ever approved by the FDA for Dravet syndrome, a form of epilepsy, and more than 500 US doctors prescribed it in November and December 2018.
That led to net sales of $4.7 million for the two-month launch period, and GW Pharmaceuticals anticipates soaring demand in the months ahead. It reported that filled prescription is up 150% in January 2019 compared to the previous month, with demand coming from major centres of excellence and local epilepsy clinics.
In December, it met with the FDA about the potential for using Sativex to treat patients with Multiple Sclerosis spasticity, and it will begin a pivotal study in Q4 2019 on this. It is also undertaking a clinical trial Tuberous Sclerosis Complex, with data expected in Q2 2019. GW Pharma has a commercial footprint in place in five major European markets, and it expects the results of its European submission for Epidiolex by Q2 2019 (learn about another company capitalizing on European markets here).
The Future of Pharmaceuticals is Bright for GW
The firm is set to enjoy continued momentum in 2019 as there is an unmet medical need to treat Dravet syndrome. It is also diversifying by launching trials into treating different conditions, and investors are impressed.
GW Pharmaceuticals Medicinal Cannabis Drug Trials
Tekla Capital Management LLC raised its holdings in GW by 45.3% during Q4, while Resources Investment Advisors Inc. and Horrell Capital Management Inc. both lifted their shares. NumerixS Investment Technologies Inc and Oppenheimer Asset Management Inc. both purchased new positions in the firm.
The share price was $97.39 on December 31, 2019, and it increased to $152.89 by February 26. It then saw a sharp increase to $173.99 after its Q4 results were announced, and it has now settled at $166.57 this week. It has a strong balance sheet, with plenty of growth potential, so it is likely to remain a popular stock among investors in the year ahead.
Recall – Not the Only Recent Stock Price Spike For GW
GW Pharmaceuticals saw its share price increase in mid-February after Alzheimer’s Research UK announced a cannabis trial with the British firm.
Cambridge-based GW Pharmaceuticals produces a cannabis mouth spray called Sativex that contains equal amounts of THC and CBD. It became the first cannabis-based medicine to be licensed in the UK and it is used to treat symptoms of multiple sclerosis, including spasticity, pain, tremors, and sleep disturbance.
Now the respected charity Alzheimer’s Research UK has decided to fund a pioneering clinical trial at King’s College in London to discover the efficacy of Sativex in treating dementia sufferers. It said that around half of the 850,000 Brits living with Alzheimer’s will experience agitation or aggression, making it one of the most challenging symptoms of the illness. It hopes that cannabis can negate this.
It is devoting around £300,000 ($384,000) to the trial, which will see a study team recruit volunteers aged between 55 and 90 who suffer from dementia and experience symptoms of aggression or agitation. They will take Sativex for four weeks and, if their symptoms improve, they will perform a much larger clinical trial.
Right now only MS patients can be prescribed Sativex, but Alzheimer’s Research UK will aim to change that if the spray proves to be effective. The researchers need to assess how practical it is to use a mouth spray if a patient is displaying extreme aggression. But chief scientific officer Dr. David Reynolds said no new dementia treatments have emerged in the past 15 years and that it is imperative for the medical community to test novel treatment methods in rigorous clinical trials.
GW Pharmaceutical’s stock price increased to $1520.50 at the close of trading on February 15th.
Cannabis is legal for medicinal purposes in the UK, but it has not taken off to the extent seen in countries like Germany. That is due to the strictness of guidelines that doctors must adhere to, but the landscape is starting to change and this trial could help unlock the industry.
The opinions provided in this article are those of the author and do not constitute investment advice. Readers should assume that the author and/or employees of Grizzle hold positions in the company or companies mentioned in the article. For more information, please see our Content Disclaimer.

